The basics of high pressure cleaning
Cleaning with high pressure is a very thorough and effective way to clean, as it even remove stubborn dirt. The use of additional cleaning agents is often unnecessary, which makes high pressure cleaning an environmentally friendly cleaning method.
What makes dirt stick?
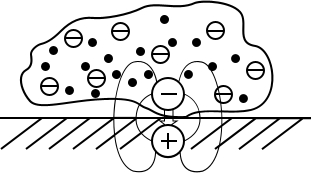
Electrostatic attraction:
If a surface is electrically charged with reverse polarity of the charge of the dirt, a bond occurs as a result of the inherent attractive forces.
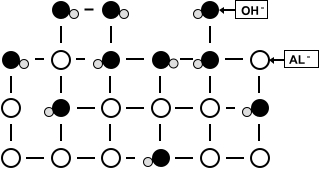
Chemical reaction:
A chemical reaction changes the composition of the surface which causes it to look dirty.
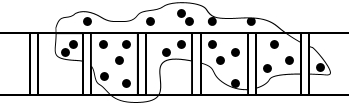
Structural interlocking:
Dirt gets caught and trapped in rough surfaces.
How are the principles of dirt adhesion applied to the cleaning process?
The aim of cleaning is to destroy the bond between the dirt and the respective surface. High pressure cleaning makes use of mechanical, thermal and chemical factors for this purpose.
Mechanical factors
There are four primary variables which affect the performance of a high pressure cleaner: flow rate, pump pressure, spraying distance and spraying angle.
Impact force, or the penetrating force of the water jet in relation to the unit of surface area to be cleaned, is most important. It depends on
- Spraying distance: a distance of 10 to 30 cm is recommended, as the pressure curve drops quickly at greater distances.
- Spraying angle: This angle should be selected according to the degree of soiling. Stubborn dirt requires an angle of 0 – 25°, medium soiling 25 – 50° and light soiling an angle of 50 – 80°.
- Nozzle pressure: as a function of water flow rate and the cross section of the nozzle, only a disproportionate increase of nozzle pressure can generate greater impact force.
- Water flow rate: a quick increase in impact force is achieved by increasing the quantity of water. This also makes it easier to remove dirt, as there is minimal atomisation even at greater distances.
Thermal factors
The addition of heat speeds up chemical processes, allowing grease and oil to be removed more quickly, for example. Because the object being cleaned is warm, the subsequent drying time is shorter too. Better results and a cleaning time that is up to 40% shorter can be achieved. However, increasing the spraying distance results in a reduction in temperature.
Chemical factors
If increasing the impact force or temperature does not produce a satisfactory outcome, then the use of chemical cleaning agents is necessary. These have a positive effect on increasing the wetting power, emulsification or direct chemical reactions with the dirt particles.
The suitability of the cleaning agents depends on their pH value and the surface being treated with them. Acidic agents with a pH value of 0-6 are used for calcium deposits, urine deposits, rust and other oxides, and alkaline agents with a pH of 8.5 to 14 are used for oils, greases, tar and soot.
Neutral cleaning solutions are the right choice for sensitive surfaces which are contaminated with small amounts of oil or grease. Extremely acidic or alkaline levels can, however, result in damage to the high pressure cleaner and the object to be cleaned itself, and may even violate water protection laws. The prolonged application of chemical cleaning agents can also cause damage. Increasing the duration of the application time is generally only effective up to certain point, as cleaning intensity approaches a threshold with the progression of time. Pre-soaking coarse layers of dirt with water can have a positive effect on cleaning time, as this can reduce the amount of cleaning time required with the high pressure cleaner by as much as 50%.






